research
The Corder Lab @ Penn investigates how the brain generates the perception of pain and aversive memories, and how dysfunction in these networks drives the transition to chronic pain and drug abuse. We combine the pharmacology of opioids and psychedelics with state-of-the-art neuroscience approaches—including in vivo imaging of neural activity, neuroanatomical circuit tracing, and optical neuromanipulation—to dissect the brain circuits and molecular mechanisms that shape pain, pleasure, and motivation.
Our current projects include:
Anterior cingulate cortex (ACC) coding of pain affect and opioid analgesia
Basolateral amygdala (BLA) connections with the insula and nucleus accumbens in regulating pain affect
Thalamocortical circuits linking sleep and pain processing
Central amygdala circuits underlying opioid dependence and withdrawal
Periaqueductal gray (PAG) circuits mediating placebo analgesia
Retrosplenial–cingulate cortical mechanisms of psilocybin and psychedelic modulation of aversive learning and memory, with relevance to PTSD and chronic pain
In addition to our core neuroscience research, we design and build innovative tools to accelerate discovery and translation. These include:
Released Markers of Activity (RMAs): enabling measurement of brain and spinal neural activity through a simple blood test.
Cell-type–specific viral promoters: providing precision access for gene therapies targeting defined neural populations.
Mu Opioid Receptor Promoters (MORp) AAV constructs from Salimando et al. Nature Comms. 2023 can be purchased from the Stanford Vector Core
AxoDen: an open-source, quantitative histology platform for automated axonal density analysis.
LUPE (Light aUtomated Pain Evaluator): a deep-learning–powered behavior analysis system that decodes complex animal behaviors to reveal how neural circuits drive pain affective-motivational states.
Our automated, preclinical behavior systems improve the accuracy and efficiency of analgesic discovery, ultimately paving the way for more effective and safer pain management strategies.
Through this integrated approach—bridging fundamental neuroscience, advanced technology development, and translational research—we aim to identify new therapeutic targets and create next-generation treatments, including precision gene therapies, to reduce mental health burdens linked to chronic pain and to lessen dependence on prescription opioids.
NEURAL CODING OF AFFECTIVE STATES
Combining machine learning with "miniscope" calcium imaging from large populations of neurons deep in the brain of behaving animals, allows us to decode the neural activities underlying the emotional component of painful and hedonic experiences.
Pain is an unpleasant experience that commands attention and the engagement of motivational protective behaviors to limit exposure to noxious stimuli. In contrast, chronic pain is not merely a persistent sensory disorder, but a neurological disease of affective dysfunction that serves no survival function. As such, chronic pain negatively impacts the mental state, professional goals, and personal relationships of over 100 million Americans. However, it is unclear how the nociceptive systems in the brain undergo pathological maladaptations to enable the transition to a chronic pain state.
Figure adapted from Corder et al. An amygdalar neural ensemble that encodes the unpleasantness of pain Science. 2019
CONTROL OF MOTIVATED BEHAVIORS DERIVED FROM FUNCTIONALLY DISTINCT NEURAL CIRCUITS
Brining together mouse genetics, viral tracing, pharmaco- and optogenetics permits the functional interrogation of neural circuits and spinal cord::brain pathways that shape complex behaviors.
In particular, painful experiences are constructed from neural information relating not only sensory, but also emotional, interoceptive, inferential, and cognitive data, which coalesce into a unified conscious perception of pain. If we can understand the processes by which these different functional dimensions of pain perceptions are generated from distinct brain networks, in particular those encoding the negative affective or unpleasantness of pain, then we can generate new dynamic frameworks for modeling the emergence of unrelenting chronic pain.
Figure adapted from Corder et al. An amygdalar neural ensemble that encodes the unpleasantness of pain Science. 2019
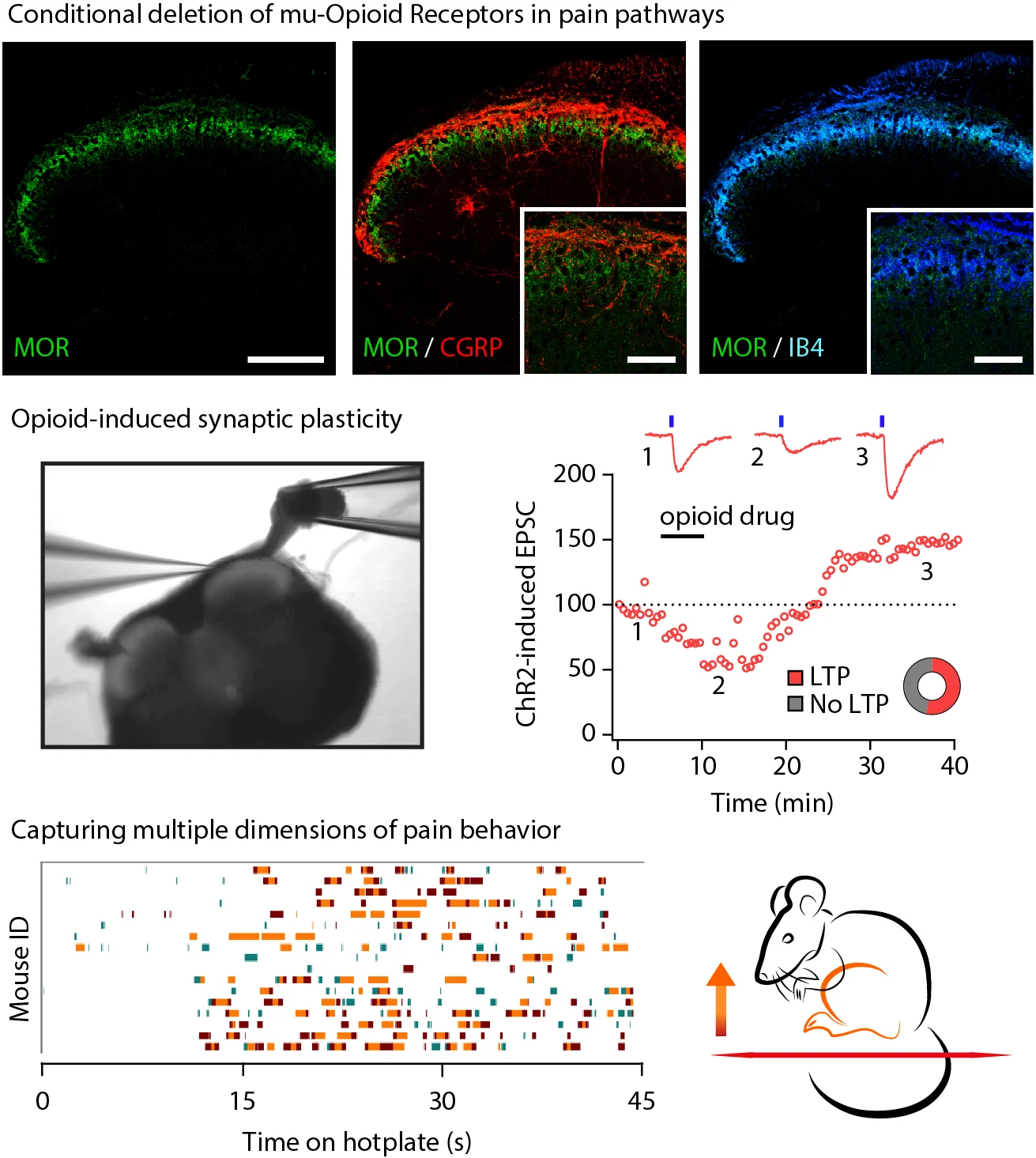
GENETIC, MOLECULAR, AND SYNAPTIC REMODELING BY OPIOIDS AND DRUGS OF ABUSE
With the staggering prevalence of chronic pain, the broad use of opioids for pain management has increased markedly in the past decades. This increase in opioid prescriptions has been accompanied by a sharp rise in the incidence of addiction and opioid-related mortality, a phenomenon termed the Opioid Epidemic. Our lab will traverse two research paths to battle the Opioid Epidemic: discovering non-opioid analgesic therapies that could replace opioids or improving current opioid analgesics. For both paths, a decisve first step must be the complete resolution of the analgesic mechanisms of opioids, at the synaptic, circuit, and network levels.
Data from Corder et al. Loss of μ opioid receptor signaling in nociceptors, but not microglia, abrogates morphine tolerance without disrupting analgesia. Nature Medicine. 2017